PCSK9 Inhibitor Market Growth Projections US$ 7.67 billion by 2033, Growth Insights (2025)
PCSK9 Inhibitor Market Demand Rising | Forecast, Drivers & Opportunities
PCSK9 Inhibitor Market Growth (2025-2033): Market Rise in North America, Japan, EU, Middle East & Africa”
AUSTIN, TX, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- Market Size and Growth 2025— DataM Intelligence 4Market Research LLP
Cardiovascular disease remains the world’s leading cause of death, and PCSK9 inhibitors are rapidly emerging as one of the most powerful next-generation therapies for lowering LDL cholesterol. With strong regulatory support, innovative biologics, and expanding clinical adoption, the global PCSK9 inhibitor is entering a high-growth phase driven by breakthrough drugs, strategic investments, and expanding treatment guidelines.
Market Overview 2025-2033:
PCSK9 Inhibitor Market was valued at US$ 1.96 billion in 2024 and is projected to reach US$ 7.67 billion by 2033, expanding at a compound annual growth rate (CAGR) of 16.5% during the forecast period from 2025 to 2033.
Download the Sample - Discover Strategic PCSK9 Market Data:- https://www.datamintelligence.com/download-sample/pcsk9-inhibitor-market
The PCSK9 inhibitor market is gaining significant traction as healthcare systems worldwide prioritize advanced treatments for hypercholesterolemia and cardiovascular disease prevention. PCSK9 inhibitors are a class of lipid-lowering drugs that block the PCSK9 protein, enabling the liver to remove more LDL cholesterol from the bloodstream and significantly reducing cardiovascular risk.
The market has experienced notable growth due to increasing cardiovascular disease prevalence and rising demand for targeted biologic therapies. Market estimates indicate that the sector is expanding rapidly and is projected to grow at a strong double-digit CAGR over the next decade as pharmaceutical companies continue investing in innovative cholesterol-management therapies.
Advances in biotechnology particularly monoclonal antibodies and RNA-interference therapies are enabling longer-acting treatments and improved patient adherence. These technological developments are positioning PCSK9 inhibitors as a key component in modern lipid management strategies.
Market Drivers: Escalating Cardiovascular Disease Burden Accelerates Demand
The rising prevalence of hypercholesterolemia and cardiovascular diseases is one of the most significant factors driving demand for PCSK9 inhibitors. Lifestyle changes, aging populations, obesity, and metabolic disorders are increasing the number of patients requiring effective cholesterol-lowering therapies.
Traditional statins remain the first-line therapy, but many patients either fail to achieve adequate LDL reduction or experience intolerance. PCSK9 inhibitors provide an alternative mechanism that can significantly lower LDL cholesterol levels and reduce cardiovascular events in high-risk populations.
In addition, growing awareness of preventive cardiology and the inclusion of PCSK9 inhibitors in clinical treatment guidelines are encouraging physicians to adopt these therapies for patients with familial hypercholesterolemia or persistent cardiovascular risk.
Market Opportunities: Next-Generation Therapies and Expanded Indications
The PCSK9 inhibitor landscape is witnessing rapid innovation, creating substantial opportunities for pharmaceutical companies and investors. New therapeutic approaches such as small interfering RNA (siRNA) therapies and oral PCSK9 inhibitors are expanding the treatment pipeline and improving patient convenience.
Moreover, research is exploring the use of PCSK9 inhibitors beyond cholesterol management, including broader cardiovascular risk reduction and precision medicine strategies. Increasing reimbursement coverage and government initiatives promoting cardiovascular health are also expected to accelerate adoption across developed and emerging healthcare markets.
Geographical Analysis: North America Leads While Asia-Pacific Accelerates
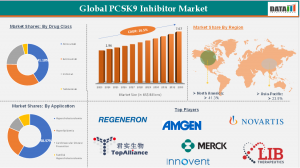
North America held the largest share of the global PCSK9 inhibitor industry, accounting for 41.3% of the total share in 2024.
North America currently holds the dominant share of the PCSK9 inhibitor market, driven by advanced healthcare infrastructure, high cardiovascular disease prevalence, strong reimbursement frameworks, and early adoption of innovative biologics.
The United States plays a critical role due to high pharmaceutical R&D investments and regulatory support for innovative lipid-lowering therapies. Meanwhile, Europe continues to expand through improved clinical guidelines and insurance coverage.
Asia-Pacific is expected to witness the fastest growth during the forecast period as countries such as Japan, China, and South Korea invest in advanced cardiovascular treatments and expand access to biologic medicines. Rising healthcare spending and growing awareness of cholesterol management are further strengthening regional demand.
Customize the Report for Deeper Competitive Insights:- https://www.datamintelligence.com/customize/pcsk9-inhibitor-market
Key Players Driving Innovation
The global PCSK9 inhibitor market is highly competitive and dominated by several leading pharmaceutical companies focusing on innovative biologics and long-acting therapies.
Key companies include:
1. Amgen
2. Sanofi
3. Regeneron Pharmaceuticals
4. Novartis
5. Merck & Co.
6. AstraZeneca
7. LIB Therapeutics
8. Shanghai Junshi Biosciences Co., Ltd,
9. Innovent Bio
10. Esperion Therapeutics
These companies are focusing on product innovation, clinical trials, strategic collaborations, and global commercialization to strengthen their market presence.
FDA Approvals of PCSK9 Inhibitors
1. Lerochol (lerodalcibep-liga) - FDA Approval (December 2025)
In December 2025, the U.S. FDA approved Lerochol (lerodalcibep-liga), a third-generation PCSK9 inhibitor developed by LIB Therapeutics.
This approval expanded the PCSK9 inhibitor class by introducing a new long-acting injectable option for cholesterol management.
2. Leqvio (inclisiran) - FDA Label Expansion (July 2025)
In July 2025, the FDA approved a new indication for Leqvio (inclisiran) from Novartis.
The approval allows Leqvio to be used earlier in the treatment pathway, improving access to PCSK9-targeting therapy for patients with high LDL cholesterol.
Inclisiran is the first RNA-interference therapy targeting PCSK9, providing sustained LDL reduction with only two doses per year.
3. Repatha (evolocumab) - Expanded FDA Indication (2025)
The FDA also expanded the indication for Repatha (evolocumab), a PCSK9 monoclonal antibody developed by Amgen.
This regulatory expansion broadened the use of PCSK9 inhibitors for earlier cardiovascular risk management, increasing the potential patient population.
Regulatory Affairs of PCSK9 Inhibitors
Regulatory frameworks across the United States, Europe, and Asia have increasingly recognized the importance of advanced lipid-lowering therapies.
Regulatory agencies require comprehensive clinical evidence demonstrating LDL reduction and cardiovascular outcome benefits before approval. Post-marketing surveillance and pharmacovigilance programs are also critical to ensuring long-term safety.
The regulatory landscape is evolving to support novel technologies such as gene-silencing therapies and precision medicine approaches, creating opportunities for innovative pharmaceutical companies.
Government Policies Supporting PCSK9 Inhibitors
Governments worldwide are implementing policies to improve cardiovascular disease prevention and management. Public health programs promoting cholesterol screening and early intervention are increasing the diagnosis of hypercholesterolemia.
In several developed countries, reimbursement policies have been expanded to support high-risk patients who require advanced lipid-lowering therapies. These initiatives are improving patient access to PCSK9 inhibitors and encouraging broader adoption within healthcare systems.
Product Launches Strengthening Market Expansion
• December 2025 - LIB Therapeutics launched Lerochol (lerodalcibep-liga), a recombinant fusion protein PCSK9 inhibitor designed to reduce LDL cholesterol in adults with hypercholesterolemia, offering sustained LDL-C reductions of around 60% in clinical trials.
• July 2025 - Amgen introduced the Repatha (evolocumab) Pushtronex system, a hands-free on-body infusor that delivers a full monthly 420 mg dose in a single administration, improving patient convenience and adherence to PCSK9 therapy.
• July 2025 - Novartis expanded the use of Leqvio (inclisiran) with an updated regulatory label allowing its use as a monotherapy with diet and exercise for LDL-C reduction, strengthening its position as a twice-yearly PCSK9-targeting treatment.
PCSK9 Inhibitor Market - Funding by Companies
1. Eli Lilly Funding & Investment in Verve Therapeutics
One of the most significant recent investments in PCSK9 gene-targeted therapies involves Eli Lilly’s strategic funding and acquisition of Verve Therapeutics.
Company: Eli Lilly
Funding Type: Strategic investment + acquisition
Investment Value:
$1 billion upfront payment
Up to $300 million milestone payments
Focus: CRISPR gene-editing therapy targeting the PCSK9 gene
The therapy VERVE-102 is designed as a single-dose gene-editing treatment that permanently switches off the PCSK9 gene to reduce LDL cholesterol levels.
2. Eli Lilly Collaboration Funding with Beam Therapeutics
Eli Lilly also funded Beam Therapeutics to support base-editing technology used in PCSK9 gene therapies.
Company: Eli Lilly
Partner: Beam Therapeutics
Funding:
$200 million upfront payment
$50 million equity investment
Purpose: Development of base-editing therapies for cardiovascular diseases including PCSK9-related cholesterol disorders.
This investment strengthened the development of precision gene-editing platforms aimed at long-term cholesterol reduction.
3. Novartis Strategic Investment in Inclisiran Development
Novartis has invested heavily in siRNA-based PCSK9 inhibition through its drug inclisiran (Leqvio).
Company: Novartis
Investment: $9.7 billion acquisition of The Medicines Company
Purpose: Development and commercialization of inclisiran, a small-interfering RNA therapy targeting PCSK9.
4. Verve Therapeutics Internal R&D Funding
Verve Therapeutics itself raised significant funding to advance PCSK9 gene-editing programs.
Company: Verve Therapeutics
Cash Position: About $539.9 million in cash and securities (2024 report)
Purpose: Funding clinical trials for VERVE-102 PCSK9 gene-editing therapy and other lipid-related programs.
This funding supports Phase 1b and Phase 2 clinical trials across multiple countries.
Buy the Full Report to Unlock Strategic Market Intelligence:- https://www.datamintelligence.com/buy-now-page?report=pcsk9-inhibitor-market
Mergers & Acquisitions Shaping the Competitive Landscape
1. Eli Lilly Acquires Verve Therapeutics (2025)
One of the most significant recent deals connected to PCSK9 therapies occurred when Eli Lilly announced the acquisition of Verve Therapeutics in a deal valued at up to US$1.3 billion.
2. Strategic Investments in RNA-Based Cardiovascular Therapies (2025-2026)
Large pharmaceutical companies are increasingly acquiring RNA-based drug platforms that include cardiovascular targets such as PCSK9.
For example, Novartis announced a US$12 billion acquisition of Avidity Biosciences, strengthening its RNA therapeutics pipeline expected to advance toward regulatory filings by 2026
3. Earlier Strategic Acquisition Supporting PCSK9 Therapies
A notable foundational acquisition shaping the current PCSK9 market landscape was:
Novartis Acquisition of The Medicines Company
Deal Value: US$9.7 billion
Asset: Inclisiran (siRNA-based PCSK9 inhibitor)
Outcome: Development and commercialization of Leqvio, a twice-yearly cholesterol-lowering therapy.
Recent Developments - United States (2025-2026)
2026 - Clinical Expansion of Next-Generation PCSK9 Therapies
Biotechnology companies in the United States advanced clinical trials for next-generation PCSK9 inhibitors designed to provide longer dosing intervals and improved cardiovascular outcomes. These innovations aim to improve treatment adherence and expand the patient population eligible for PCSK9 therapy.
2025 - Investment in RNA-Interference-Based PCSK9 Treatments
Pharmaceutical companies increased investments in RNA-interference technology targeting PCSK9 pathways. This approach focuses on reducing PCSK9 production in the liver and delivering long-lasting cholesterol reduction with fewer doses.
Recent Developments - Japan (2025-2026)
2026 - Expansion of PCSK9 Therapy Access in Japan
Japanese healthcare institutions expanded the adoption of PCSK9 inhibitors for high-risk cardiovascular patients following updated clinical guidelines and improved reimbursement frameworks.
2025 - Clinical Research Collaborations on Lipid-Lowering Biologics
Japanese pharmaceutical companies and research institutions strengthened collaborations to develop advanced PCSK9 inhibitors and improve treatment outcomes for patients with familial hypercholesterolemia and severe hyperlipidemia.
Conclusion: A High-Growth Therapeutic Segment in Cardiovascular Medicine
The PCSK9 inhibitor market is entering a transformative growth phase driven by the increasing burden of cardiovascular disease, technological innovation in biologics, and supportive regulatory frameworks.
With pharmaceutical companies investing heavily in research, product innovation, and strategic collaborations, PCSK9 inhibitors are poised to become a cornerstone therapy in advanced cholesterol management. As healthcare systems focus on preventive cardiology and precision medicine, the market is expected to deliver significant opportunities for pharmaceutical leaders, investors, and healthcare stakeholders worldwide.
Related Reports
PI3K Inhibitor Drug Class Market
Enzyme Inhibitor Market
Sai Kiran
DataM Intelligence 4market Research LLP
+1 877-441-4866
sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.